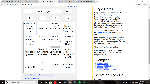
4.18J/g*K
Overall ΔH = stepwise ΔH Used to measure of flow of heat T of surrounding increases work done by system ΔE= Ef - Ei = ΔE+PΔV = q =Hprod- Hreact products and reactants Ability to do work or transfer heat equal to -PΔV specific heat of aqueous sol equal to -qsol ; Cs*msol*ΔT -Ccal*ΔT Heat is absorbed by the system Amount of energy to raise T of 1n by 1K water and beaker Heat or Enthalpy of Reaction 4.18J/g*K Overall ΔH = stepwise ΔH Used to measure of flow of heat T of surrounding increases work done by system ΔE= Ef - Ei = ΔE+PΔV = q =Hprod- Hreact products and reactants Ability to do work or transfer heat equal to -PΔV specific heat of aqueous sol equal to -qsol ; Cs*msol*ΔT -Ccal*ΔT Heat is absorbed by the system Amount of energy to raise T of 1n by 1K water and beaker Heat or Enthalpy of Reaction
(Print)
4.18J/g*K
Overall ΔH
= stepwise ΔH
Used to measure of flow of heat
T of surrounding increases
work done by system
ΔE= Ef - Ei
= ΔE+PΔV
= q
=Hprod-Hreact
products and reactants
Ability to do work or transfer heat
equal to
-PΔV
specific heat of aqueous sol
equal to
-qsol ;
Cs*msol*ΔT
-Ccal*ΔT
Heat is absorbed by the system
Amount of energy to raise T of 1n by 1K
water
and beaker
Heat or Enthalpy of Reaction