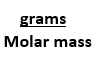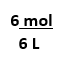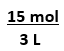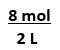Electrolyte High Specific Heat Dissociation Super- saturated Non- polar NON Electrolyte Equation has 2 numbers, g--> mol, mL --> L (s) insoluble solvent Heat, Stir, Reduce Pressure negative slope M1 x V1 = M2 x V2 1500.0 mL = 1.5 L Surface Area miscible Polar solution (aq) soluble 30.0 mL = 0.03 L Saturated High boiling point Equation has 3 numbers, solve for X Dilution Equation Heat, Stir, Crush 1000.0 mL = 1 L Low Vapor Pressure immiscible positive slope Solubility graph Y axis All nitrates are soluble Solubility unsaturated High Surface Tension Solubility graph X axis 750.0 mL = 0.75 L solute Homogeneous solution 500.0 mL = 0.5 L Electrolyte High Specific Heat Dissociation Super- saturated Non- polar NON Electrolyte Equation has 2 numbers, g--> mol, mL --> L (s) insoluble solvent Heat, Stir, Reduce Pressure negative slope M1 x V1 = M2 x V2 1500.0 mL = 1.5 L Surface Area miscible Polar solution (aq) soluble 30.0 mL = 0.03 L Saturated High boiling point Equation has 3 numbers, solve for X Dilution Equation Heat, Stir, Crush 1000.0 mL = 1 L Low Vapor Pressure immiscible positive slope Solubility graph Y axis All nitrates are soluble Solubility unsaturated High Surface Tension Solubility graph X axis 750.0 mL = 0.75 L solute Homogeneous solution 500.0 mL = 0.5 L
(Print)
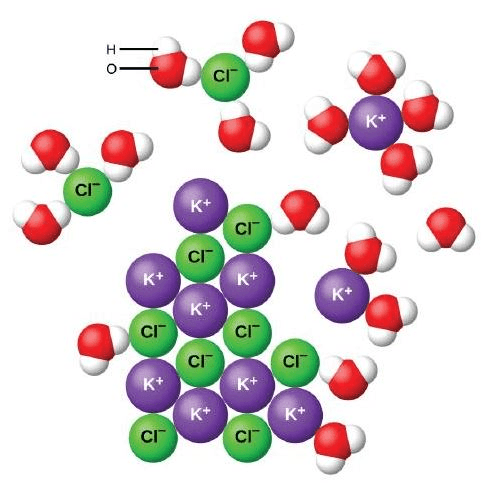
Electrolyte
High Specific Heat
Dissociation
Super-saturated
Non-polar
NON
Electrolyte
Equation has 2 numbers,
g--> mol,
mL --> L
(s) insoluble
solvent
Heat,
Stir, Reduce Pressure
negative slope
M1 x V1
=
M2 x V2
1500.0
mL = 1.5 L
Surface Area
miscible
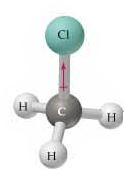
Polar
solution
(aq) soluble
30.0
mL = 0.03 L
Saturated
High boiling point
Equation has 3 numbers, solve for X
Dilution Equation
Heat,
Stir, Crush
1000.0
mL
= 1 L
Low Vapor Pressure
immiscible
positive slope
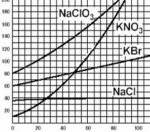
Solubility graph
Y axis
All nitrates are soluble
Solubility
unsaturated
High Surface Tension
Solubility graph
X axis
750.0
mL = 0.75 L
solute
Homogeneous solution
500.0
mL = 0.5 L