Photoelectric Effect Oil Drop Experiment Formulated the "Atomic Theory of Matter" Planetary Model of the Atom Uncertainty Principle Wave Particle Duality Democritus Law of Definite Proportions Moseley Use of Mass Spectroscopy to discover isotopes Thomson the "Law of Conservation of Matter" Seaborg Planck Newlands Mendeleev Chadwick Rutherford's Atomic Model Schrodinger Joseph Proust Coulomb's Law Photoelectric Effect Oil Drop Experiment Formulated the "Atomic Theory of Matter" Planetary Model of the Atom Uncertainty Principle Wave Particle Duality Democritus Law of Definite Proportions Moseley Use of Mass Spectroscopy to discover isotopes Thomson the "Law of Conservation of Matter" Seaborg Planck Newlands Mendeleev Chadwick Rutherford's Atomic Model Schrodinger Joseph Proust Coulomb's Law
(Print)
Photoelectric Effect
Oil Drop Experiment
Formulated the "Atomic Theory of Matter"
Planetary Model of the Atom
Uncertainty Principle
Wave Particle Duality
Democritus
Law of Definite Proportions
Moseley
Use of Mass Spectroscopy to discover isotopes
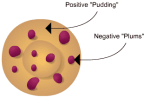
Thomson
the "Law of Conservation of Matter"
Seaborg
Planck
Newlands
Mendeleev
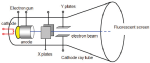
Chadwick
Rutherford's Atomic Model
Schrodinger
Joseph Proust
Coulomb's Law