invert SN2 stereochemistry E2 elimination bimolecular SN2 reaction E2 example E2, E1 favors most substituted alkene isomer racemicize/ epimerize stereocenter SN1 stereochemistry:? polar aprotic SN2 solvent:? Free! E1 example ROH, NH3, H20 E1 solvents primary/ secondary SN2 Carbon:? polar protic SN1 solvent:? lone pair SN1 and E1 nucleophile:? E1 elimination unimolecular SN1 reaction SN1 substitution nucleophile unimolecular E1, SN1 Which reactions go through carbocation? SN1, E2, E1 tertiary or secondary? elimination make alkenes negative charge SN2 and E2 nucleophile:? SN2 substitution nucleophile bimolecular regioisomers the same functional groups but attached at different positions not important E2 solvent? invert SN2 stereochemistry E2 elimination bimolecular SN2 reaction E2 example E2, E1 favors most substituted alkene isomer racemicize/ epimerize stereocenter SN1 stereochemistry:? polar aprotic SN2 solvent:? Free! E1 example ROH, NH3, H20 E1 solvents primary/ secondary SN2 Carbon:? polar protic SN1 solvent:? lone pair SN1 and E1 nucleophile:? E1 elimination unimolecular SN1 reaction SN1 substitution nucleophile unimolecular E1, SN1 Which reactions go through carbocation? SN1, E2, E1 tertiary or secondary? elimination make alkenes negative charge SN2 and E2 nucleophile:? SN2 substitution nucleophile bimolecular regioisomers the same functional groups but attached at different positions not important E2 solvent?
(Print)
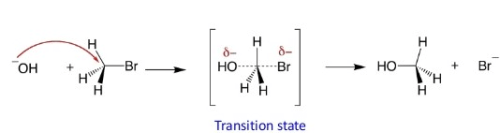
SN2 stereochemistry
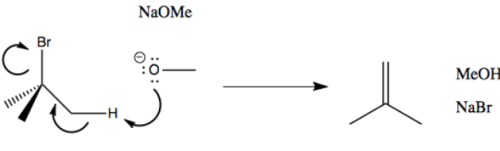
elimination bimolecular
SN2 reaction
E2 example
favors most substituted alkene isomer
SN1 stereochemistry:?
SN2 solvent:?
Free!
E1 example
E1 solvents
SN2 Carbon:?
SN1 solvent:?
SN1 and E1 nucleophile:?
elimination unimolecular
SN1 reaction
substitution
nucleophile
unimolecular
Which reactions go through carbocation?
tertiary or secondary?
make alkenes
SN2 and E2 nucleophile:?
substitution
nucleophile
bimolecular
the same functional groups
but attached at different positions
E2 solvent?