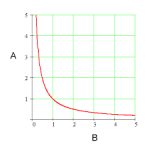
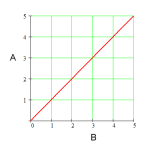
9atm V1/T1 = V2/T2 doubled 900 K Free! True low temp & high pressure Hydrogen & Helium Kinetic Molecular Theory (KMT) P1V1/T1 = P2V2/T2 P1V1 = P2V2 Xe lighter 48 L Boyle's Law stays the same Kelvin Molecules 819 K False halved 298 K 22.4 L 6.02 x 10^23 101.3 kPa 139 mL 546 K high temp & low pressure 9atm V1/T1 = V2/T2 doubled 900 K Free! True low temp & high pressure Hydrogen & Helium Kinetic Molecular Theory (KMT) P1V1/T1 = P2V2/T2 P1V1 = P2V2 Xe lighter 48 L Boyle's Law stays the same Kelvin Molecules 819 K False halved 298 K 22.4 L 6.02 x 10^23 101.3 kPa 139 mL 546 K high temp & low pressure
(Print)
B-9atm
G-V1/T1 = V2/T2
N-doubled
I-900 K
B-Free!
I-True
G-low temp & high pressure
G-Hydrogen & Helium
I-Kinetic Molecular Theory (KMT)
B-P1V1/T1 = P2V2/T2
B-P1V1 = P2V2
I-Xe
I-lighter
N-48 L
N-
N-Boyle's Law
N-stays the same
I-Kelvin
I-Molecules
N-819 K
B-False
B-
G-halved
N-298 K
G-22.4 L
B-6.02 x 10^23
B-101.3 kPa
I-139 mL
G-546 K
G-high temp & low pressure